Divetalking » Adv. Open Water, Article, Dive Planner, Divetalking, Education, Enriched Air, Featured, NItrox, RDP, Recreational Dive Planner, Reference, Report, Specialties, Training » Understanding Enriched Air Nitrox
Understanding Enriched Air Nitrox
September 3rd, 2020 | Comments Off on Understanding Enriched Air Nitrox

Let me start the article with stating what you are about to read is NOT a substitute for training from a certified Enriched Air Instructor. What you read here compliments that instruction. It may be used to prep before you participate in instruction or as a refresher. Enriched Air Certification is earned only through a certified Enriched Air Instructor. Do Not attempt to dive Nitrox without being certified.
If you are interested in becoming Enriched Air certified, known as Nitrox throughout this document feel free to reach out to Larry Davis or your local dive organization.
There will be a vast variety of experiences reading this article. The intent of the article is for someone wanting to know more about Nitrox. Since the age, level of experience, knowledge of the individual reading is not known the article is written to allow the least knowledged and experience person to learn. This may prove too basic for your level of understanding so please keep this in mind when reading.
You may be here because you are curious and you want to learn. When you complete the article, you will be more knowledgeable and not at a superficial level. If your intention is to take a course after going through this article, I am confident that when you understand the concepts, know the formulas and have ingrained images that reinforce the Why’s, How’s, When, and What’s you will be more prepared for the course and it’s quizzes as well as having become a more knowledgeable and safer diver.
The way the article is written is to make one think. Concept are introduced with the intent to implant a thought. Questions may be asked that may read a little off. You may find questions being asked with no immediate answer provided. This too is intended to introduce concepts and terms. When you come across these types of questions, make note of them. We will be returning to the point later in the article. It may just be too soon for the details.
As the article progresses acronyms, concepts and terms introduced earlier will begin to be defined and be used more often. You may feel a bit overwhelmed at times if a subject is something new to you. My advise, read it again, perhaps take it in smaller chunks and if necessary to clarify an understanding, reach out to the author. Larry is an excellent instructor and will help when he can. The intent is to introduce and reinforce concepts and points, not to make someone feel incompetent.
We all know reading something without having access to immediate clarification may be a bit taunting. I understand there may be those who will pick and point out deviations and challenge them. For example.. throughout the article we will refer to the percentage of Oxygen and Nitrogen in a scuba cylinder filled with air as 21% and 79% respectively. We know this not to be true. Oxygen and Nitrogen are approximately 20.8xy% and 78.6xy% respectively. For simplicity Oxygen and Nitrogen have been rounded to 21% and 79%. We’re not looking at sending someone to Mars, just out of this world diving.
Before you begin. I recommend you take the Nitrox Quiz. After reading the article, take the quiz again and compare the two quizzes to see your knowledge gained.
Click HERE to take the quiz
What is Nitrox?
The air you breath is primarily comprised of Oxygen and Nitrogen along with minuscule amounts of other gasses such as argon, carbon dioxide, neon, methane, helium, krypton, hydrogen, xenon, ozone, nitrogen dioxide, iodine, carbon monoxide, and ammonia. For the purpose of this discussion those other minuscule amounts of others gasses will be forgotten as we focus on the two primary gasses, Oxygen and Nitrogen.
It is simplest terms, Nitrox is the addition of extra Oxygen added to your scuba cylinder as a substitute for Nitrogen. The result is a tank that has a higher percentage of Oxygen and a lower percentage of Nitrogen when compared to a tank filled with air. Recreational Nitrox cylinders may be filled with Oxygen between 21.01% and 40%.
Nitrox is simply substituting a percentage of the Nitrogen with Oxygen.
The two scuba cylinders below have one filled with air which has 21% Oxygen and 79% nitrogen and a Nitrox cylinder filled with 32% Oxygen will have 68% Nitrogen. The sum of the percentage of gasses in a bottle must equal 100%. Another way of saying this is the Fraction of Oxygen, FO in the tank to the left is .21 and the FO in the tank to the right is .32. Make note of the term Fraction of Oxygen, FO. We will be using it later.

AIR 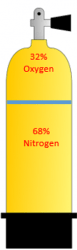
32% EAN
Would it be fair to say the total pressure exerted by the two gasses, part of that total pressure comes from Oxygen and the other Nitrogen? Yes, of course it would. We could elaborate the total pressure is Partially from the FO in the cylinder and partially from the Fraction of Nitrogen, FN for a combined pressure of 100%.
© 2020 – 2021, admin. All rights reserved.
Pages: 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
Filed under: Adv. Open Water, Article, Dive Planner, Divetalking, Education, Enriched Air, Featured, NItrox, RDP, Recreational Dive Planner, Reference, Report, Specialties, Training · Tags: diving on nitrox, EAN, Enriched air, enriched air nitrox, learn about nitrox, Nitrox, Nitrox certification, Understanding nitrox, Viewport, what is enriched air, What is nitrox
Log Log and Calculator
Cloud Tag
Most commented
Recent Articles
Tag Cloud
artificial reef blowing up reefs Canon 5D Mark III cay sal conservation cozumel death dive diver dive show divetalking dolphin explosive reefs FIsh ID fishinar Fred Morrow Fred Morrow Artificial Reef Fred Morrow Reef FWC Leatherback lionfish lion fish lionfish derby lionfish harvesting logo lost missing Narcosis Nekton Ocean Photography polution Preservation RDP recall Recreational Dive Planner reef scuba show Sharks Shark Weak titanic tuna Underwater Photography Workshop Zeagle© 2026 Divetalking








